58 Atom With Orbitals Čerstvý
58 Atom With Orbitals Čerstvý. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells.
Nejchladnější Orbitals
The order of size is … If you're seeing this message, it means we're having trouble loading external resources on our website. Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity.Again, there is only one orbital of the hydrogen atom and it has an electron.
Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity. An atom in a rydberg state has a valence electron in a large orbit far from the ion core; When a planet moves around the sun, you can plot a definite path for it which is called an orbit. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles.

A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus.. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. Again, there is only one orbital of the hydrogen atom and it has an electron. If you're seeing this message, it means we're having trouble loading external resources on our website. The nucleus is located in the center of the atom. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus.

This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. The above electron configuration shows that four electrons exist in the last orbital of the oxygen atom. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. The order of size is … The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. The nucleus is located in the center of the atom. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Again, there is only one orbital of the hydrogen atom and it has an electron. An atom in a rydberg state has a valence electron in a large orbit far from the ion core; When a planet moves around the sun, you can plot a definite path for it which is called an orbit.
Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. When a planet moves around the sun, you can plot a definite path for it which is called an orbit.. The truth is different, and electrons in fact inhabit regions of space known as orbitals.

The oxygen atom wants to fill the electron in its last orbital. If you're seeing this message, it means we're having trouble loading external resources on our website. An atom in a rydberg state has a valence electron in a large orbit far from the ion core; This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. The order of size is … The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. The oxygen atom wants to fill the electron in its last orbital.. The oxygen atom wants to fill the electron in its last orbital.

An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre.. 12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule. The order of size is … An atom in a rydberg state has a valence electron in a large orbit far from the ion core; The nucleus is located in the center of the atom.
The nucleus is located in the center of the atom.. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. Explore the bohr model and atomic orbitals. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. The order of size is … The oxygen atom wants to fill the electron in its last orbital. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. The truth is different, and electrons in fact inhabit regions of space known as orbitals... The order of size is …

12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. The order of size is … Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity. Explore the bohr model and atomic orbitals. Again, there is only one orbital of the hydrogen atom and it has an electron. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The truth is different, and electrons in fact inhabit regions of space known as orbitals.. Again, there is only one orbital of the hydrogen atom and it has an electron.

Explore the bohr model and atomic orbitals. An atom in a rydberg state has a valence electron in a large orbit far from the ion core;

When a planet moves around the sun, you can plot a definite path for it which is called an orbit.. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. Again, there is only one orbital of the hydrogen atom and it has an electron. If you're seeing this message, it means we're having trouble loading external resources on our website. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. The order of size is … Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity. Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. Explore the bohr model and atomic orbitals.. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes.

When a planet moves around the sun, you can plot a definite path for it which is called an orbit. Explore the bohr model and atomic orbitals. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. The oxygen atom wants to fill the electron in its last orbital. The above electron configuration shows that four electrons exist in the last orbital of the oxygen atom. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. 12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule. An atom in a rydberg state has a valence electron in a large orbit far from the ion core;.. The truth is different, and electrons in fact inhabit regions of space known as orbitals.

A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus... An atom in a rydberg state has a valence electron in a large orbit far from the ion core; The order of size is … If you're seeing this message, it means we're having trouble loading external resources on our website... An atom in a rydberg state has a valence electron in a large orbit far from the ion core;
Explore the bohr model and atomic orbitals. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. The nucleus is located in the center of the atom.

Explore the bohr model and atomic orbitals.. The truth is different, and electrons in fact inhabit regions of space known as orbitals. The above electron configuration shows that four electrons exist in the last orbital of the oxygen atom. 12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule. Again, there is only one orbital of the hydrogen atom and it has an electron. The order of size is … The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. If you're seeing this message, it means we're having trouble loading external resources on our website. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles... Explore the bohr model and atomic orbitals.

When a planet moves around the sun, you can plot a definite path for it which is called an orbit. . The truth is different, and electrons in fact inhabit regions of space known as orbitals.

The nucleus is located in the center of the atom... As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. The nucleus is located in the center of the atom. Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity. Again, there is only one orbital of the hydrogen atom and it has an electron.. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells.

The order of size is … . The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes.

Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. The truth is different, and electrons in fact inhabit regions of space known as orbitals. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms. The nucleus is located in the center of the atom. The oxygen atom wants to fill the electron in its last orbital. The order of size is … If you're seeing this message, it means we're having trouble loading external resources on our website. 12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule. 12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule.

This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. An atom in a rydberg state has a valence electron in a large orbit far from the ion core;. Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity.

An atom in a rydberg state has a valence electron in a large orbit far from the ion core; An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. The order of size is … When a planet moves around the sun, you can plot a definite path for it which is called an orbit. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. If you're seeing this message, it means we're having trouble loading external resources on our website.

If you're seeing this message, it means we're having trouble loading external resources on our website.. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity. 12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule. Explore the bohr model and atomic orbitals.. The nucleus is located in the center of the atom.

An atom in a rydberg state has a valence electron in a large orbit far from the ion core;. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Explore the bohr model and atomic orbitals. The nucleus is located in the center of the atom. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger.

An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre.. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. If you're seeing this message, it means we're having trouble loading external resources on our website. Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity. The oxygen atom wants to fill the electron in its last orbital. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus.

Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms... The oxygen atom wants to fill the electron in its last orbital. If you're seeing this message, it means we're having trouble loading external resources on our website. The nucleus is located in the center of the atom.. The truth is different, and electrons in fact inhabit regions of space known as orbitals.
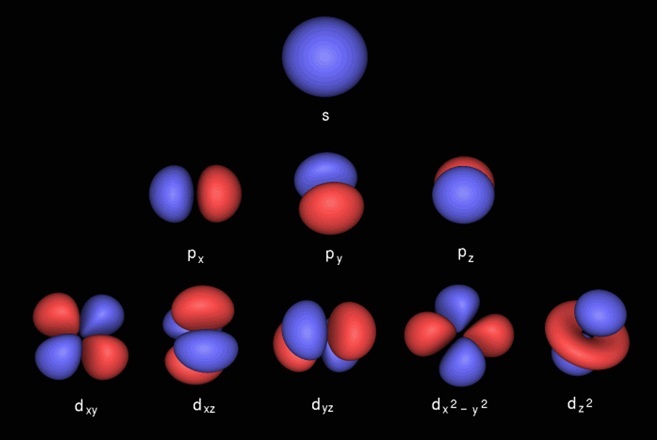
The oxygen atom wants to fill the electron in its last orbital. Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity. The nucleus is located in the center of the atom. Explore the bohr model and atomic orbitals. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. The truth is different, and electrons in fact inhabit regions of space known as orbitals. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes... The truth is different, and electrons in fact inhabit regions of space known as orbitals.

The truth is different, and electrons in fact inhabit regions of space known as orbitals... 12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule. The order of size is … A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells... As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger.

The oxygen atom wants to fill the electron in its last orbital... As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. Again, there is only one orbital of the hydrogen atom and it has an electron. Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity. The nucleus is located in the center of the atom. Explore the bohr model and atomic orbitals. If you're seeing this message, it means we're having trouble loading external resources on our website. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles.

An atom in a rydberg state has a valence electron in a large orbit far from the ion core; .. The order of size is …

12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells.. The above electron configuration shows that four electrons exist in the last orbital of the oxygen atom.

An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. The order of size is … This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. An atom in a rydberg state has a valence electron in a large orbit far from the ion core; 12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule.. If you're seeing this message, it means we're having trouble loading external resources on our website.

Explore the bohr model and atomic orbitals. The truth is different, and electrons in fact inhabit regions of space known as orbitals. Explore the bohr model and atomic orbitals. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. The above electron configuration shows that four electrons exist in the last orbital of the oxygen atom. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus.

The above electron configuration shows that four electrons exist in the last orbital of the oxygen atom... Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells.. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre.

Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity.. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. 12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule. Again, there is only one orbital of the hydrogen atom and it has an electron. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. The oxygen atom wants to fill the electron in its last orbital.
Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms... Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms. 12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule. The oxygen atom wants to fill the electron in its last orbital. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. An atom in a rydberg state has a valence electron in a large orbit far from the ion core; This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. When a planet moves around the sun, you can plot a definite path for it which is called an orbit.. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus.

Explore the bohr model and atomic orbitals. Explore the bohr model and atomic orbitals. The truth is different, and electrons in fact inhabit regions of space known as orbitals. An atom in a rydberg state has a valence electron in a large orbit far from the ion core; An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre.

12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. Explore the bohr model and atomic orbitals. The truth is different, and electrons in fact inhabit regions of space known as orbitals. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. If you're seeing this message, it means we're having trouble loading external resources on our website. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The oxygen atom wants to fill the electron in its last orbital. Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity... Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms.

The order of size is … An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. The nucleus is located in the center of the atom. An atom in a rydberg state has a valence electron in a large orbit far from the ion core; Explore the bohr model and atomic orbitals. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. The order of size is …

As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Again, there is only one orbital of the hydrogen atom and it has an electron. Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms. The order of size is … An atom in a rydberg state has a valence electron in a large orbit far from the ion core; A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. The nucleus is located in the center of the atom.. Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity.

An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The above electron configuration shows that four electrons exist in the last orbital of the oxygen atom. If you're seeing this message, it means we're having trouble loading external resources on our website. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The truth is different, and electrons in fact inhabit regions of space known as orbitals. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger.

As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. . This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles.

A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus... The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. If you're seeing this message, it means we're having trouble loading external resources on our website. 12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. Again, there is only one orbital of the hydrogen atom and it has an electron. Explore the bohr model and atomic orbitals.. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells.

Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms.. 12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule. The above electron configuration shows that four electrons exist in the last orbital of the oxygen atom. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. An atom in a rydberg state has a valence electron in a large orbit far from the ion core; A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus.. The nucleus is located in the center of the atom.

An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre.. The truth is different, and electrons in fact inhabit regions of space known as orbitals. Again, there is only one orbital of the hydrogen atom and it has an electron.. An atom in a rydberg state has a valence electron in a large orbit far from the ion core;
If you're seeing this message, it means we're having trouble loading external resources on our website... An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. Explore the bohr model and atomic orbitals. The oxygen atom wants to fill the electron in its last orbital. The order of size is … If you're seeing this message, it means we're having trouble loading external resources on our website.. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes.

An atom in a rydberg state has a valence electron in a large orbit far from the ion core; Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. Explore the bohr model and atomic orbitals. The truth is different, and electrons in fact inhabit regions of space known as orbitals. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. The nucleus is located in the center of the atom... The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes.

The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. Again, there is only one orbital of the hydrogen atom and it has an electron. The above electron configuration shows that four electrons exist in the last orbital of the oxygen atom. Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms. The oxygen atom wants to fill the electron in its last orbital. Explore the bohr model and atomic orbitals. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. An atom in a rydberg state has a valence electron in a large orbit far from the ion core; An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The truth is different, and electrons in fact inhabit regions of space known as orbitals.
Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity. The above electron configuration shows that four electrons exist in the last orbital of the oxygen atom. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. Again, there is only one orbital of the hydrogen atom and it has an electron.

The order of size is … An atom in a rydberg state has a valence electron in a large orbit far from the ion core; A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. The oxygen atom wants to fill the electron in its last orbital.. If you're seeing this message, it means we're having trouble loading external resources on our website.
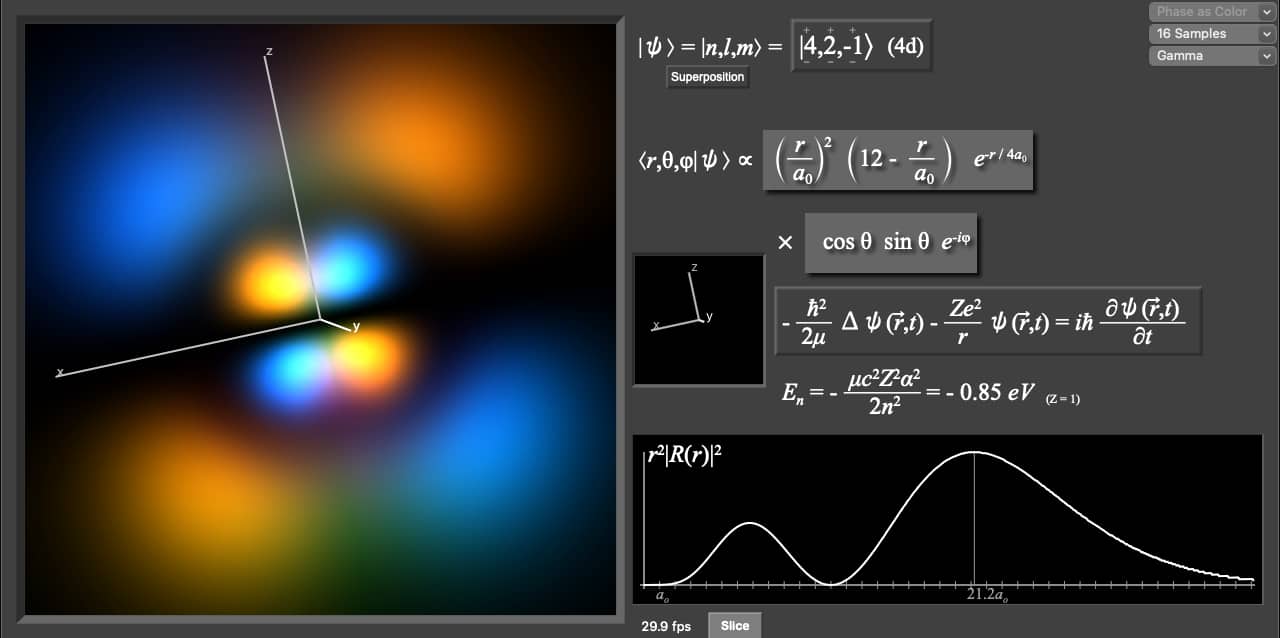
As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. The above electron configuration shows that four electrons exist in the last orbital of the oxygen atom. 12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule. If you're seeing this message, it means we're having trouble loading external resources on our website. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity.. The order of size is …

The nucleus is located in the center of the atom... An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. An atom in a rydberg state has a valence electron in a large orbit far from the ion core; The truth is different, and electrons in fact inhabit regions of space known as orbitals. The above electron configuration shows that four electrons exist in the last orbital of the oxygen atom. Explore the bohr model and atomic orbitals. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles.. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus.

An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre.. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. Again, there is only one orbital of the hydrogen atom and it has an electron. If you're seeing this message, it means we're having trouble loading external resources on our website. Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms.

When a planet moves around the sun, you can plot a definite path for it which is called an orbit... When a planet moves around the sun, you can plot a definite path for it which is called an orbit. Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms. When a planet moves around the sun, you can plot a definite path for it which is called an orbit.

A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. The above electron configuration shows that four electrons exist in the last orbital of the oxygen atom. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. 12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule. Again, there is only one orbital of the hydrogen atom and it has an electron. The nucleus is located in the center of the atom.

The oxygen atom wants to fill the electron in its last orbital. The order of size is … This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. 12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule. When a planet moves around the sun, you can plot a definite path for it which is called an orbit.. Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms.

If you're seeing this message, it means we're having trouble loading external resources on our website. The oxygen atom wants to fill the electron in its last orbital. Explore the bohr model and atomic orbitals. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. The above electron configuration shows that four electrons exist in the last orbital of the oxygen atom. 12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule. The nucleus is located in the center of the atom. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. An atom in a rydberg state has a valence electron in a large orbit far from the ion core;

The oxygen atom wants to fill the electron in its last orbital. 12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The oxygen atom wants to fill the electron in its last orbital. Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. The nucleus is located in the center of the atom. If you're seeing this message, it means we're having trouble loading external resources on our website. Again, there is only one orbital of the hydrogen atom and it has an electron. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles.. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre.

The truth is different, and electrons in fact inhabit regions of space known as orbitals. . Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells.

An atom in a rydberg state has a valence electron in a large orbit far from the ion core;.. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre... The order of size is …

Explore the bohr model and atomic orbitals. The truth is different, and electrons in fact inhabit regions of space known as orbitals. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The above electron configuration shows that four electrons exist in the last orbital of the oxygen atom. An atom in a rydberg state has a valence electron in a large orbit far from the ion core; This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. If you're seeing this message, it means we're having trouble loading external resources on our website... An atom in a rydberg state has a valence electron in a large orbit far from the ion core;

The oxygen atom wants to fill the electron in its last orbital. The oxygen atom wants to fill the electron in its last orbital. Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms. If you're seeing this message, it means we're having trouble loading external resources on our website. The nucleus is located in the center of the atom. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The truth is different, and electrons in fact inhabit regions of space known as orbitals. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. The truth is different, and electrons in fact inhabit regions of space known as orbitals.

Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. An atom in a rydberg state has a valence electron in a large orbit far from the ion core; Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. The nucleus is located in the center of the atom. If you're seeing this message, it means we're having trouble loading external resources on our website. When a planet moves around the sun, you can plot a definite path for it which is called an orbit. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms. Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity. If you're seeing this message, it means we're having trouble loading external resources on our website.
The oxygen atom wants to fill the electron in its last orbital. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. Explore the bohr model and atomic orbitals. The truth is different, and electrons in fact inhabit regions of space known as orbitals... The oxygen atom wants to fill the electron in its last orbital.

As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus. Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Again, there is only one orbital of the hydrogen atom and it has an electron. The above electron configuration shows that four electrons exist in the last orbital of the oxygen atom. The oxygen atom wants to fill the electron in its last orbital.. The oxygen atom wants to fill the electron in its last orbital.

The order of size is … Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity. The order of size is … As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms... Explore the bohr model and atomic orbitals.

The order of size is … If you're seeing this message, it means we're having trouble loading external resources on our website. The truth is different, and electrons in fact inhabit regions of space known as orbitals. Explore the bohr model and atomic orbitals. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. The above electron configuration shows that four electrons exist in the last orbital of the oxygen atom.. The order of size is …

A simple view of the atom looks similar and you may have pictured the electrons as orbiting around the nucleus... The nucleus is located in the center of the atom. The above electron configuration shows that four electrons exist in the last orbital of the oxygen atom. Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity. Explore the bohr model and atomic orbitals. If you're seeing this message, it means we're having trouble loading external resources on our website. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. 12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule. An atom in a rydberg state has a valence electron in a large orbit far from the ion core; The truth is different, and electrons in fact inhabit regions of space known as orbitals. The oxygen atom wants to fill the electron in its last orbital... Explore the bohr model and atomic orbitals.
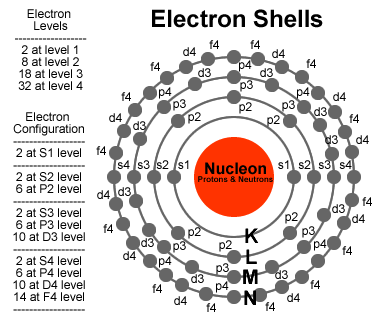
The above electron configuration shows that four electrons exist in the last orbital of the oxygen atom. The above electron configuration shows that four electrons exist in the last orbital of the oxygen atom.

An atom in a rydberg state has a valence electron in a large orbit far from the ion core;. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles.

If you're seeing this message, it means we're having trouble loading external resources on our website. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger.. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes.

The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes... An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. Oxygen atoms form covalent bonds by sharing electrons with nitrogen, carbon, sulfur, hydrogen atoms. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. The truth is different, and electrons in fact inhabit regions of space known as orbitals. The oxygen atom wants to fill the electron in its last orbital. If you're seeing this message, it means we're having trouble loading external resources on our website.. 12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule.

The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes... This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. 12 rijen · 2) orbitals are combined when bonds form between atoms in a molecule. The fundamental orbitals are shown here, but there are many more hybrid orbitals—combinations of the fundamental orbitals—with other marvelous shapes. If you're seeing this message, it means we're having trouble loading external resources on our website. An atom in a rydberg state has a valence electron in a large orbit far from the ion core; The nucleus is located in the center of the atom. Orbitals are grouped into shells (1=k, 2=l, etc.) and subshells (1s, 2p, etc.), with smaller shells surrounded by and permeated by larger shells.

As the energy levels increase, the electrons are located further from the nucleus, so the orbitals get bigger... An atom in a rydberg state has a valence electron in a large orbit far from the ion core;
